
This organization of each electron in an atom explains the shape of the periodic table but it also helps in understanding that every element in the same column will show similar properties. For example, H, Li, Na, K, Rb, and Cs all have a similar electronic configuration, which is 1s1. Now if one looks at the valence shell's electronic configuration, then it can be seen that every column has similar valence shells' electronic configuration. Hence, the first two rows in the periodic table are named s-block, while the one on the right side is named as p-block. There are ten elements with this configuration that appear in the center of the periodic table. Once 4s is filled, the 3d subshell will get occupied. After this, the electrons don't go in 3d instead they will go in 4s. 
And after this, the 3p subshell will get filled by six elements. 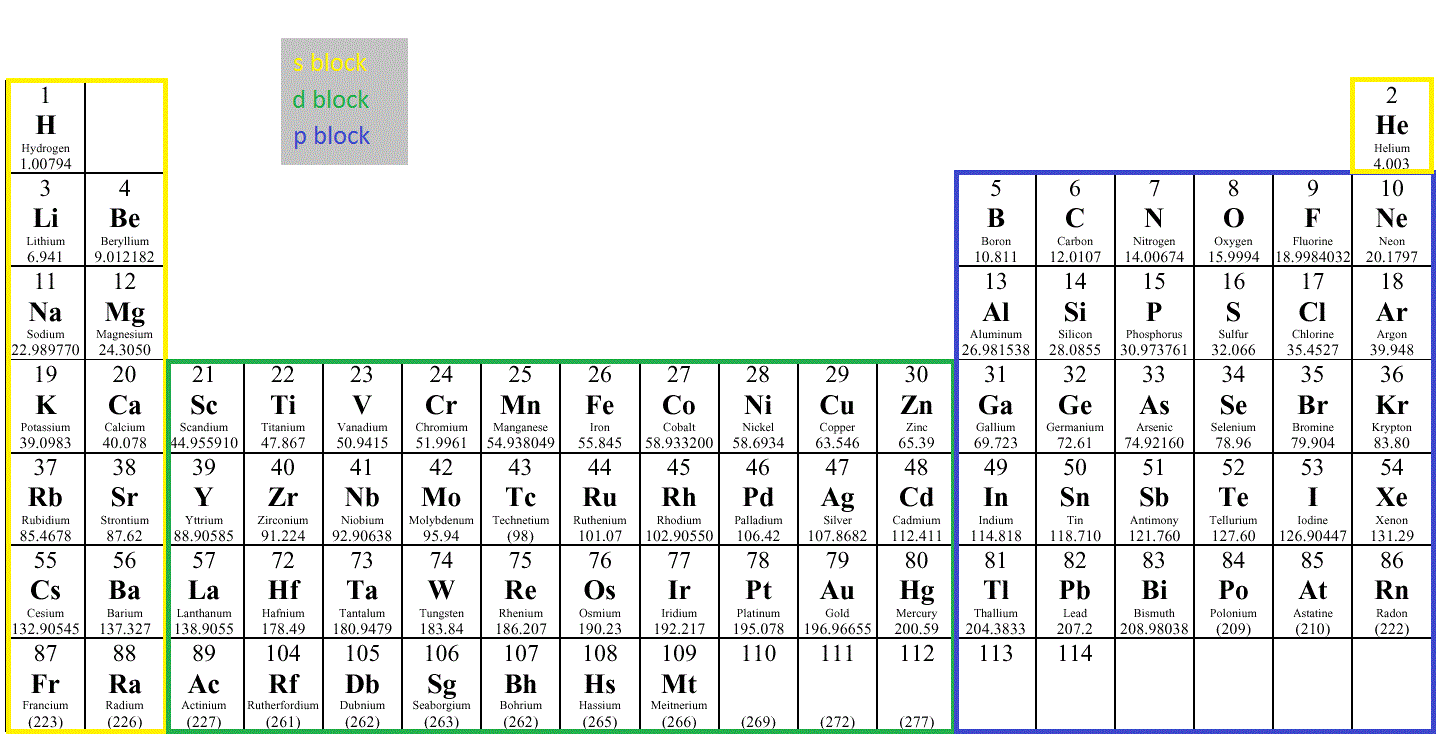
Next is the 3s subshell which is present in Na and Mg, that appear on the left side. The next in the periodic table are the six elements that occupy the 2p subshell which appears on the right side of the periodic table (from B to Ne). The next element in the periodic table is lithium and beryllium which have the 2s subshell. These two elements occupy the first row in the periodic table. Consider Hydrogen and Helium which have 1s1 and 1s2Įlectronic configurations. The shape of the periodic table is so because it resembled the filling of subshells with electrons. This can be understood by studying the electronic configurations. Now the structure of the periodic table is designed in a specific way. In a periodic table, elements are listed by using their atomic numbers. The formula is 2 n 2 \rm 1 s 2 2 s 2 2 p 6 3 s 2 3 p 5 By general formula, it can be determined how many electrons can a shell holder. The electron configurations are written in the noble gas notation. There can be only two-electron in the first shell, while the second shell can contain up to 8 electrons, also there is a tendency shown by the third shell to hold only 18 electrons, and this goes on. This periodic table contains each elements atomic number, atomic mass, symbol, name, and electron configuration. Every shell has a specific allocation of electrons. Electron configurations can be predicted by the position of an atom. Similarly, they are also represented by using alphabets like K, L, M. The arrangement of electrons in atoms is responsible for the shape of the periodic table. These shells are named like this as they correspond to the principal quantum number (Hence, n = 1, 2, 3,…). This is followed by M shell or3-shell and thus it keeps going on. The shell following this is L shell or 2-shell. The shell which is closest to the nucleus is called a K shell or is represented as 1-shell.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
